Cell Systems | Glycemia shifts pancreatic islet rhythmicity by influencing interactions between δ cells and α cells
Blood glucose homeostasis is fundamental to maintaining normal metabolism, and pancreatic islets, as the core functional units regulating blood glucose, rely heavily on intercellular communication and collective coordination. Islets are composed of approximately 80% β cells (which secrete insulin), 15% α cells (which secrete glucagon), and 5% δ cells (which secrete somatostatin), arranged in close proximity to form a complex paracrine regulatory network. Functionally, islets exhibit rhythmic calcium oscillations that drive pulsatile hormone secretion.
Recent studies have confirmed that rhythmic islet oscillations are essential for blood glucose control. However, one key question has long remained unresolved: the human pancreas contains millions of islets (and mice contain several thousand), so how do they coordinate like an orchestra to maintain stable blood glucose? Existing studies have been limited to single islets or ex vivo tissues, lacking the technical means to synchronously observe the dynamic behavior of dozens or even hundreds of islets in living organisms. Therefore, how hundreds or thousands of islets collectively “shift gears” to sense and encode changes in blood glucose—from high to normal, and from stable to fluctuating states—remains to be elucidated.
Recently, the team led by Huixia Ren at the Institute of Physiology, Capital Medical Science Innovation Center, in collaboration with the Hangzhou Institute of Medicine, Chinese Academy of Sciences, Peking University, Shenzhen University, and others, published a study in Cell Systems entitled Glycemia shifts pancreatic islet rhythmicity by influencing interactions between δ cells and α cells. Using an islet-specific fluorescent mouse model together with a wide-field imaging system for continuous glucose monitoring, the researchers achieved, for the first time, dynamic calcium imaging of 20–100 islets in the pancreas of live mice for more than 4 hours. They found that systemic blood glucose levels are encoded by the rhythmic calcium oscillations of the islet population, and that the core regulatory network underlying these rhythms consists of the minority islet cell types, δ cells and α cells (Figure 1).
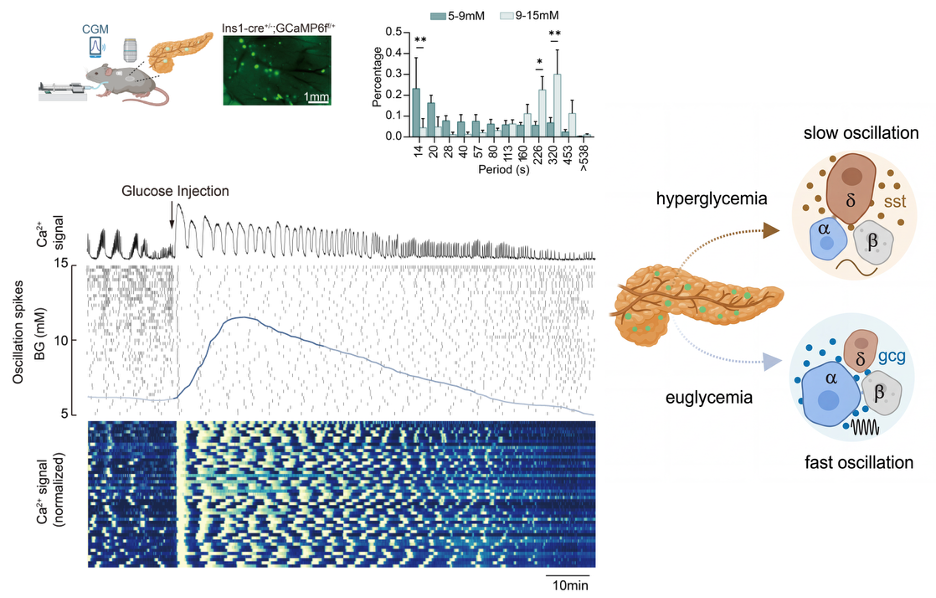
Figure 1: To investigate how blood glucose regulates islet population activity, the research team achieved synchronous calcium imaging of 20–100 islets in live mice. They found that islet calcium oscillation rhythms undergo a precise and coordinated slow-to-fast transition with changing blood glucose levels: when blood glucose falls from a hyperglycemic state back to the normal range, the oscillation period synchronously switches from a slow cycle of 226–320 seconds to a fast cycle of 14–20 seconds. This transition is an important feature by which healthy islets adapt to glucose changes and maintain glucose homeostasis.
Based on this phenomenon of“slow-to-fast rhythmic switching” during the transition from hyperglycemia back to euglycemia (Hyperglycemia to Euglycemia induced Slow to Fast oscillation, HESF), the team further designed experiments to investigate its regulatory mechanism and its alteration in disease models:
1. Multilevel models reveal that glucose-dependent rhythm switching requires “dialogue” within islets
To verify the regulatory basis of this phenomenon, the researchers established a multilevel experimental model consisting of in vivo glucose clamp, pancreatic tissue slices, and dispersed single cells (Figure 2). The results showed that glucose-dependent rhythm switching was still present in pancreatic tissue slices, where paracrine signaling between cells is preserved, but completely disappeared in dispersed β cells, where paracrine communication is lost. This clearly demonstrates that intercellular paracrine signaling within islets is the core regulatory basis of the HESF phenomenon, expanding beyond the previous view that focused only on β-cell autonomous rhythms.
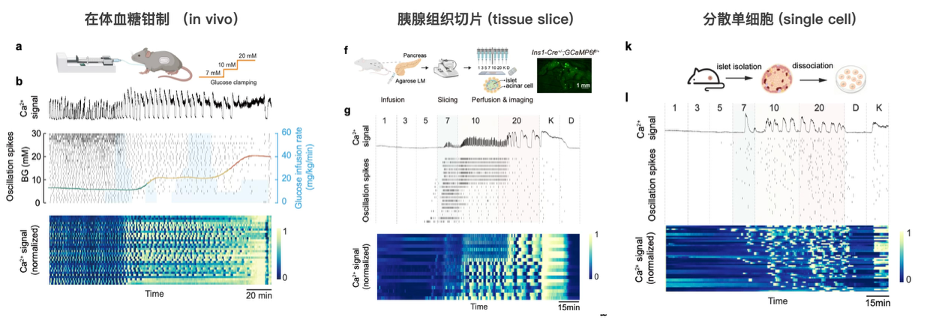
Figure 2: Multilevel experimental model consisting of in vivo glucose clamp, pancreatic tissue slices, and dispersed single cells.*
2. Mechanistic dissection of islet rhythm regulation: interactions between δ cells and α cells dominate islet rhythmic control
To explore the paracrine mechanisms underlying islet rhythmicity, the researchers performed high-glucose stimulation experiments in isolated islets in vitro (Figure 3). Under high-glucose conditions, islets exhibited two intrinsic and reproducible calcium oscillation patterns—fast oscillations and slow oscillations—allowing the islets to be divided into two categories. Further experiments showed that adding exogenous glucagon (secreted by α cells) converted both fast- and slow-oscillating islets into the fast oscillation mode, whereas adding somatostatin (secreted by δ cells) converted both types into the slow oscillation mode. These findings indicate that interactions between δ cells and α cells play a critical role in regulating islet rhythmicity: **activation of α cells drives fast oscillations, whereas activation of δ cells maintains slow oscillations.
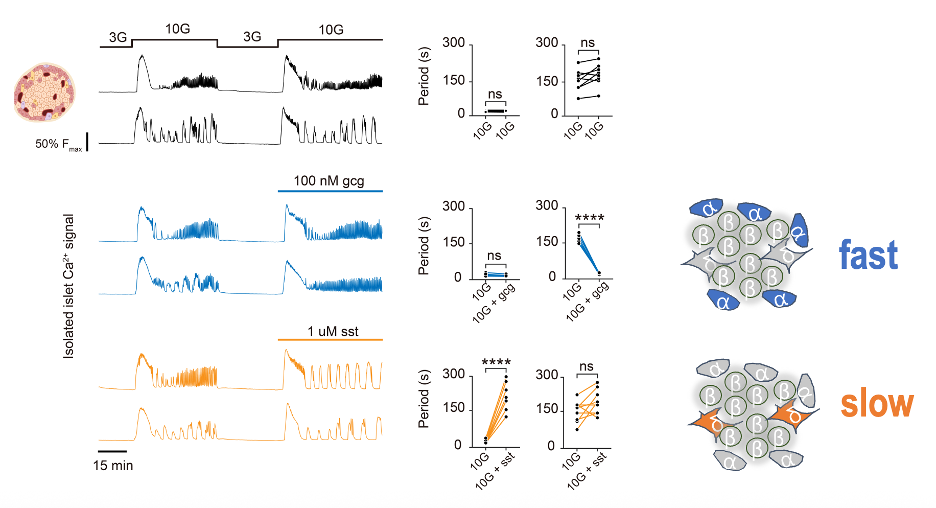
Figure 3: Exogenous glucagon and somatostatin alter oscillation modes in isolated islets.*
3. Mechanism of glucose encoding: δ–α cell interactions constitute the core of islet rhythm regulation
How, then, do blood glucose levels achieve rhythmic encoding through interactions between δ cells and α cells? Previous studies have shown that α cells respond strongly to low glucose, whereas δ cells respond strongly to high glucose. Based on this, the team proposed the following model: under hyperglycemic conditions, δ cells are activated and secrete somatostatin, which suppresses α-cell activity and causes islets to exhibit slow calcium oscillations; when blood glucose returns to normal, the inhibitory effect of δ cells is relieved, α cells are activated and secrete glucagon, thereby triggering fast calcium oscillations in islets. To further test this hypothesis, the team carried out receptor-blocking experiments. The results showed that blocking the inhibition of α cells by δ cells, or activating the relevant signaling pathways, could effectively regulate the calcium oscillation period of islets (Figure 4). These experiments confirm that δ–α cell interactions are the core mechanism by which blood glucose states are encoded into islet rhythms: α-cell activation drives fast oscillations, δ-cell activation drives slow oscillations, and the two together mediate dynamic responses and rhythmic switching through paracrine signaling.
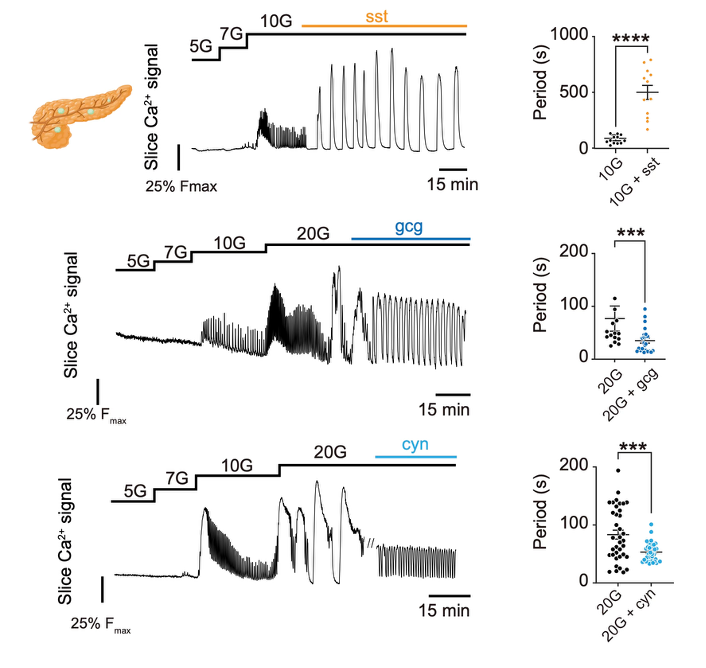
Figure 4: Exogenous glucagon and somatostatin alter glucose-induced islet oscillation modes in tissue slices.
4. A key defect in diabetes: loss of islet rhythm-switching capacity
To investigate the relationship between this regulatory mechanism and diabetes, the team conducted validation experiments using obese diabetic (ob/ob) mice (Figure 5). The results showed that diabetic mice exhibited marked disruption of blood glucose homeostasis, and their islets completely lost the HESF rhythm-switching capacity both in vivo and in vitro. Specifically, they failed to generate fast calcium oscillations even under normal glucose conditions, and the slow oscillations seen under high glucose also lost their glucose-dependent regulation. At the same time, islet sensitivity to changes in glucose was significantly reduced, as reflected by abnormal activation at low glucose levels and reduced sustained activation under high glucose. These defects are important contributors to impaired glucose regulation in diabetic mice.
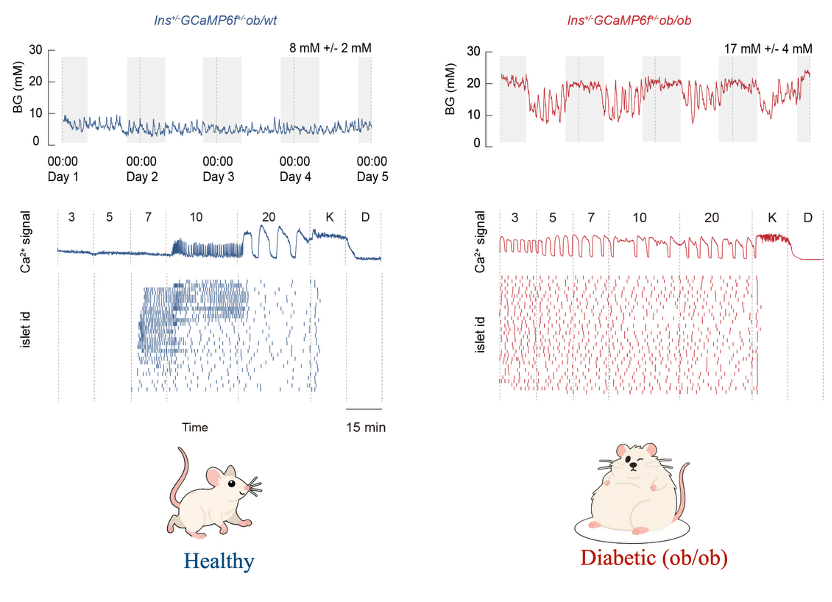
Figure 5: Continuous glucose monitoring in healthy and diabetic mice (top) and calcium activity of islet populations under glucose stimulation (bottom).
5. Islet rhythm switching depends on activation of the intra-islet GLP-1 receptor
Mechanistically, the researchers found that the generation of fast calcium oscillations in islets depends on endogenous activation of the GLP-1 receptor (Glp1r) (Figure 6). The GLP-1 receptor agonist semaglutide effectively activated this pathway, not only rapidly lowering blood glucose in diabetic mice, but also restoring their HESF rhythm-switching capacity and bringing islet calcium oscillation patterns close to those seen in normal mice. Figure 6: Perturbation of the GLP-1 receptor activation pathway alters in vivo islet calcium oscillation modes and restores blood glucose regulation and calcium rhythmicity in diabetic mice.
Summary
This study reveals, at the in vivo level, a new mechanism by which blood glucose regulates islet rhythmicity, and demonstrates the central role of δ–α cell interaction-mediated islet rhythm regulation in hyperglycemia encoding. This provides a new perspective for understanding how blood glucose homeostasis is maintained. At the same time, the study shows that loss of islet rhythm-switching capacity is an important feature of dysregulated glucose control in diabetes, and that GLP-1 receptor activation can effectively restore this function. These findings offer a new direction for diabetes treatment and lay the experimental foundation for developing new drugs targeting δ–α cell interactions.
This work was supported by the Capital Medical Science Innovation Center, the National Natural Science Foundation of China, the Hangzhou Institute of Medicine of the Chinese Academy of Sciences, and multiple other projects and platforms.
Doctoral student Yawen Deng, postdoctoral researcher Zhenchao Fu, and doctoral student Xuejiao Wang from the Capital Medical Science Innovation Center are co-first authors of the paper. Huixia Ren from the Capital Medical Science Innovation Center, Yuanyuan Du from the Hangzhou Institute of Medicine of the Chinese Academy of Sciences, and Xiaohong Peng from Shenzhen University are co-corresponding authors.
Watch the video introducion at https://www.youtube.com/watch?v=ls4Cbtv8k90
