News
Here you see our lab activies, including attending an academic conference, presenting their research and lab outing …
2026

Blood glucose homeostasis is fundamental to maintaining normal metabolism, and pancreatic islets, as the core functional units regulating blood glucose, rely heavily on intercellular communication and collective coordination. Islets are composed of approximately 80% β cells (which secrete insulin), 15% α cells (which secrete glucagon), and 5% δ cells (which secrete somatostatin), arranged in close proximity to form a complex paracrine regulatory network. Functionally, islets exhibit rhythmic calcium oscillations that drive pulsatile hormone secretion. Recent studies have confirmed that rhythmic islet oscillations are essential for blood glucose control. However, one key question has long remained unresolved: the human pancreas contains millions of islets (and mice contain several thousand), so how do they coordinate like an orchestra to maintain stable blood glucose? Existing studies have been limited to single islets or ex vivo tissues, lacking the technical means to synchronously observe the dynamic behavior of dozens or even hundreds of islets in living organisms. Therefore, how hundreds or thousands of islets collectively “shift gears” to sense and encode changes in blood glucose—from high to normal, and from stable to fluctuating states—remains to be elucidated. Recently, the team led by Huixia Ren at the Institute of Physiology, Capital Medical Science Innovation Center, in collaboration with the Hangzhou Institute of Medicine, Chinese Academy of Sciences, Peking University, Shenzhen University, and others, published a study in Cell Systems entitled Glycemia shifts pancreatic islet rhythmicity by influencing interactions between δ cells and α cells. Using an islet-specific fluorescent mouse model together with a wide-field imaging system for continuous glucose monitoring, the researchers achieved, for the first time, dynamic calcium imaging of 20–100 islets in the pancreas of live mice for more than 4 hours. They found that systemic blood glucose levels are encoded by the rhythmic calcium oscillations of the islet population, and that the core regulatory network underlying these rhythms consists of the minority islet cell types, δ cells and α cells (Figure 1). Figure 1: To investigate how blood glucose regulates islet population activity, the research team achieved synchronous calcium imaging of 20–100 islets in live mice. They found that islet calcium oscillation rhythms undergo a precise and coordinated slow-to-fast transition with changing blood glucose levels: when blood glucose falls from a hyperglycemic state back to the normal range, the oscillation period synchronously switches from a slow cycle of 226–320 seconds to a fast cycle of 14–20 seconds. This transition is an important feature by which healthy islets adapt to glucose changes and maintain glucose homeostasis. Based on this phenomenon of“slow-to-fast rhythmic switching” during the transition from hyperglycemia back to euglycemia (Hyperglycemia to Euglycemia induced Slow to Fast oscillation, HESF), the team further designed experiments to investigate its regulatory mechanism and its alteration in disease models: 1. Multilevel models reveal that glucose-dependent rhythm switching requires “dialogue” within islets To verify the regulatory basis of this phenomenon, the researchers established a multilevel experimental model consisting of in vivo glucose clamp, pancreatic tissue slices, and dispersed single cells (Figure 2). The results showed that glucose-dependent rhythm switching was still present in pancreatic tissue slices, where paracrine signaling between cells is preserved, but completely disappeared in dispersed β cells, where paracrine communication is lost. This clearly demonstrates that intercellular paracrine signaling within islets is the core regulatory basis of the HESF phenomenon, expanding beyond the previous view that focused only on β-cell autonomous rhythms. Figure 2: Multilevel experimental model consisting of in vivo glucose clamp, pancreatic tissue slices, and dispersed single cells.* 2. Mechanistic dissection of islet rhythm regulation: interactions between δ cells and α cells dominate islet rhythmic control To explore the paracrine mechanisms underlying islet rhythmicity, the researchers performed high-glucose stimulation experiments in isolated islets in vitro (Figure 3). Under high-glucose conditions, islets exhibited two intrinsic and reproducible calcium oscillation patterns—fast oscillations and slow oscillations—allowing the islets to be divided into two categories. Further experiments showed that adding exogenous glucagon (secreted by α cells) converted both fast- and slow-oscillating islets into the fast oscillation mode, whereas adding somatostatin (secreted by δ cells) converted both types into the slow oscillation mode. These findings indicate that interactions between δ cells and α cells play a critical role in regulating islet rhythmicity: **activation of α cells drives fast oscillations, whereas activation of δ cells maintains slow oscillations. Figure 3: Exogenous glucagon and somatostatin alter oscillation modes in isolated islets.* 3. Mechanism of glucose encoding: δ–α cell interactions constitute the core of islet rhythm regulation How, then, do blood glucose levels achieve rhythmic encoding through interactions between δ cells and α cells? Previous studies have shown that α cells respond strongly to low glucose, whereas δ cells respond strongly to high glucose. Based on this, the team proposed the following model: under hyperglycemic conditions, δ cells are activated and secrete somatostatin, which suppresses α-cell activity and causes islets to exhibit slow calcium oscillations; when blood glucose returns to normal, the inhibitory effect of δ cells is relieved, α cells are activated and secrete glucagon, thereby triggering fast calcium oscillations in islets. To further test this hypothesis, the team carried out receptor-blocking experiments. The results showed that blocking the inhibition of α cells by δ cells, or activating the relevant signaling pathways, could effectively regulate the calcium oscillation period of islets (Figure 4). These experiments confirm that δ–α cell interactions are the core mechanism by which blood glucose states are encoded into islet rhythms: α-cell activation drives fast oscillations, δ-cell activation drives slow oscillations, and the two together mediate dynamic responses and rhythmic switching through paracrine signaling. Figure 4: Exogenous glucagon and somatostatin alter glucose-induced islet oscillation modes in tissue slices. 4. A key defect in diabetes: loss of islet rhythm-switching capacity To investigate the relationship between this regulatory mechanism and diabetes, the team conducted validation experiments using obese diabetic (ob/ob) mice (Figure 5). The results showed that diabetic mice exhibited marked disruption of blood glucose homeostasis, and their islets completely lost the HESF rhythm-switching capacity both in vivo and in vitro. Specifically, they failed to generate fast calcium oscillations even under normal glucose conditions, and the slow oscillations seen under high glucose also lost their glucose-dependent regulation. At the same time, islet sensitivity to changes in glucose was significantly reduced, as reflected by abnormal activation at low glucose levels and reduced sustained activation under high glucose. These defects are important contributors to impaired glucose regulation in diabetic mice. Figure 5: Continuous glucose monitoring in healthy and diabetic mice (top) and calcium activity of islet populations under glucose stimulation (bottom). 5. Islet rhythm switching depends on activation of the intra-islet GLP-1 receptor Mechanistically, the researchers found that the generation of fast calcium oscillations in islets depends on endogenous activation of the GLP-1 receptor (Glp1r) (Figure 6). The GLP-1 receptor agonist semaglutide effectively activated this pathway, not only rapidly lowering blood glucose in diabetic mice, but also restoring their HESF rhythm-switching capacity and bringing islet calcium oscillation patterns close to those seen in normal mice. Figure 6: Perturbation of the GLP-1 receptor activation pathway alters in vivo islet calcium oscillation modes and restores blood glucose regulation and calcium rhythmicity in diabetic mice. Summary This study reveals, at the in vivo level, a new mechanism by which blood glucose regulates islet rhythmicity, and demonstrates the central role of δ–α cell interaction-mediated islet rhythm regulation in hyperglycemia encoding. This provides a new perspective for understanding how blood glucose homeostasis is maintained. At the same time, the study shows that loss of islet rhythm-switching capacity is an important feature of dysregulated glucose control in diabetes, and that GLP-1 receptor activation can effectively restore this function. These findings offer a new direction for diabetes treatment and lay the experimental foundation for developing new drugs targeting δ–α cell interactions. This work was supported by the Capital Medical Science Innovation Center, the National Natural Science Foundation of China, the Hangzhou Institute of Medicine of the Chinese Academy of Sciences, and multiple other projects and platforms. Doctoral student Yawen Deng, postdoctoral researcher Zhenchao Fu, and doctoral student Xuejiao Wang from the Capital Medical Science Innovation Center are co-first authors of the paper. Huixia Ren from the Capital Medical Science Innovation Center, Yuanyuan Du from the Hangzhou Institute of Medicine of the Chinese Academy of Sciences, and Xiaohong Peng from Shenzhen University are co-corresponding authors. Watch the video introducion at https://www.youtube.com/watch?v=ls4Cbtv8k90

On February 9, 2026, lab members gathered together to celebrate the Spring Festival of the Year of the Horse. We set up 11 distinctive awards, and all members voted to select the top vote-getter for each award, with joy filling the award presentation session. Winners received red envelopes for the New Year. We enjoyed the steaming lamb spine feast on site made the atmosphere particularly warm. Laughter and joy abounded throughout the gathering. In a relaxed and pleasant atmosphere, we embraced the new year’s research journey with full enthusiasm.
2025

Dec. 18, 2025, PhD candidates Wang Xuejiao and Yang Sen successfully defended their thesis proposals at the Ren Huixia Laboratory. Wang Xuejiao’s research topic is titled ‘The function of pancreatic delta cells in health and diabetes: early links to reduced endogenous somatostatin release’. Yang Sen’s thesis proposal focuses on the divergent roles of α- and δ-cells in tuning pancreatic islet glucose sensitivity during glycemic elevation and reduction. These defense sessions marked a key milestone in their doctoral research journeys, with their proposed studies promising to provide valuable insights into pancreatic cell biology and diabetes-related mechanisms. Congratulations!

The “Brain, Body & Behavioral Homeostasis” conference was held in Shenzhen from December 4th to 7th, 2025. Homeostasis is the prerequisite for life and health. Abnormal regulation of innate behaviors such as defense, eating and sleep leads to impaired homeostasis. Leading experts shared the latest scientific insights and technology development in sleep homeostasis, brain control of body homeostasis and brain-body interactions. In an open and exploratory academic environment, the speaker and the participants engaged in constructive and in-depth discussion, jointly expanding the cognitive boundaries of this cutting-edge field. Lab members Dr. Huixia Ren, Dr. Zhenchao Fu and student Yawen Deng attended the conference. Yawen introduced her study in poster. Dr. Huixia Ren delivered a talk titled: Hyperglycemia to euglycemia shift pancreatic islets rhythmicity in vivo, impairment in diabetes.

From November 19 to 22, 2025, Dr. Liu Zhimei and Li Dan attended the 27th Academic Conference of the Chinese Diabetes Society, which was held in Xi’an. Focusing on the latest research advances and clinical practices in the field of diabetes, the conference covered a wide range of topics including basic research, clinical application, as well as the prevention and management of diabetes and its complications. Participants shared cutting-edge progress in diabetes prevention and treatment.

Beijing, Nov. 7-8, 2025-To strengthen inter-lab collaboration and take a break from research, members of Ren Lab and Wu Zhaofa Lab at the Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, went on a two-day autumn trip to Qinglong Gorge in Huairou District, Beijing. The group walked along canyon trails, admired the autumn scenery with glistening water and colorful foliage, explored the nearby Great Wall, and tried the thrilling bungee jumping. On the first night, they enjoyed a hot pot feast and a cozy karaoke session together. This trip helped researchers unwind, deepen mutual understanding, and lay a solid foundation for future interdisciplinary cooperation.

Vienna, Austria – September 15–19, 2025. The Ren Laboratory participated in the 61st Annual Meeting of the European Association for the Study of Diabetes (EASD), held at the Vienna Congress & Convention Center. The congress brought together almost 14,000 international researchers, clinicians, and healthcare professionals to exchange the latest advances in diabetes research and care.

CIMR held the 2025 Annual Retreat from August 27 to August 29, 2025, at Sunshine International Conference Center, Beijing. Huixia Ren and all lab members attended the annual retreat. They prepared posters to communicate with the teachers about their research results. Prof. Kim Sneppen was invited to gaved an presentation titled “A rule based physical model for morphological diversity”. This retreat strengthen interdisciplinary cooperation and exchanges in biomedicine.

From August 26–28, 2025, we co-organized the “Digital Life Forum: Trans-Scale Imaging and Integrative Islet Biology” in Huairou Science City, Beijing. The forum brought together global experts to discuss innovations in imaging and islet biology, with a focus on diabetes research.

On Aug. 22th, 2025, “Metabolic Biology - Cell Press Joint Conference & The 8th Academic Symposium of the Metabolic Biology Branch of the Chinese Biophysical Society” was successfully held in Wenzhou, Zhejiang from August 21st to 25th, 2025. Dr. Huixia Ren was invited to attend and gave a sharing report titled “Hyperglycemia to euglycemia shift pancreatic islets rhythmicity in vivo, impairment in diabetes”, while her students Sen Yang and Xuejiao Wang also attended the conference. They all engaged in academic exchanges with domestic and foreign peers to enhance the laboratory’s research influence in the field of metabolic biology.

Beijing, June 5 – We hold a seminar on “Mesoscopic Microscopy Imaging” featuring Dr. Shi Guohua from the Hangzhou Institute of Medicine, Chinese Academy of Sciences. His talk reviewed the development and current status of this cutting‑edge high‑resolution, large‑field imaging technology, which was named one of the top 10 physics breakthroughs in 2016. Dr. Shi presented his latest research on multi‑wavelength mesoscale objectives and imaging applications, and discuss future prospects.

Pisa, Italy – The European Association for the Study of Diabetes (EASD) host its 1st Early Career Academy Meeting (ECAM) from April 28 to 30, 2025. The event aims to shape the future of diabetes research and care by fostering collaboration among early career scientists and clinicians. Participants are engaged in cutting‑edge discussions, network with leading experts, and explore innovative approaches to tackling diabetes. This first edition in Pisa was a vibrant platform for sharing research, building partnerships, and inspiring the next generation of diabetes researchers.

“Membrane Proteins: Physiology to Pharmacology” conference successfully took place at the Cold Spring Harbor Asia Academic Center in Suzhou from April 14th to 18th, 2025. The conference attracted top scientists and researchers from around the globe to explore the physiological functions and structural characteristics of membrane proteins. Lab members Dr. Huixia Ren, Xuejiao Wang and Sen Yang attended the conference. We exhibited our laboratory’ past and ongoing studies via poster. Xuejiao Wang and Sen Yang introduced the association between the Ca2+ signal and islets’ function to the audience who are interested in our poster. Dr. Huixia Ren contributed an oral presentation.
2024

December 25, 2024 – We invited Chief Physician Shusen Wang from Tianjin First Central Hospital. Dr. Wang presented recent advances in stem cell‑derived islet transplantation, a promising therapy for type 1 diabetes that addresses the shortage of donor islets. The talk highlighted research published in Cell and Nature Medicine, demonstrating how lab‑grown islets can restore insulin function in humans and primates.

Beijing, Nov. 4, 2024 – Lab members from Ren Lab and the Wu Zhaofa Lab at the Institute of Genetics, Chinese Academy of Sciences, embarked on a joint autumn trip to Gubei Water Town. The group explored the Simatai Great Wall, experienced traditional folk activities such as tie‑dyeing, and enjoyed the picturesque autumn scenery. The outing provided a relaxed setting for scientists from both labs to interact, share ideas, and build new friendships. The event strengthened interdisciplinary connections and fostered a spirit of collaboration beyond the laboratory.

On July 1st, 2024, RenLab started at CIMR (Chinese Institutes for Medical Research, Beijing). Welcome to join us!

On June 18h, the European Islet Study Group 2024 Meeting was held in Helsinki, Finland. Dr. Huixia Ren attended the conference with an invited talk. The meeting brings together 250 researchers from all over the world. And everyone enjoy the white northern summer nights!

On April 19th, 2024, Prof. Patrick Gilon from Biomedical Sciences at the Catholic University of Louvain (UCL) visit Chen Lab and Tang Lab at Peking University. During his visit, he gave a seminar about “Direct and indirect mechanisms of control of glucagon secretion” and had lunch with students. He is the Director of Research of the Fonds National de la Recherche Scientifique (Belgium). His main interests are to study stimulation secretion-coupling of insulin- and glucagon-secreting cells and interactions between the different cell types of the endocrine pancreas in normal and physiopathological conditions linked to diabetes. One major part of his research was to study the mechanisms of control of the free Ca2+ concentration in various subcellular compartments. He has published approximately 80 articles on these topics.

On Mar. 7th, Brain & Endocrine: Back to the Basics conference was held in Hong Kong. Dr. Huixia Ren gave an oral presentation “δ-α cell-to-cell interactions modulate pancreatic islet Ca²⁺ oscillation modes”. In this conference, leading experts shared their latest scientific insights into the intricate mechanisms behind the release of brain signals and hormones in response to external stimuli, as well as how these signals shape our behavior and metabolism. Explore cutting-edge research on the interplay between neuronal circuits and hormone pathways, and their impact on diverse aspects of physiology and disease.
2023

On July 29th, the 17th Q-Bio conference took place in Shen Zhen, China. Dr. Huixia Ren contributed to an oral presentation. The annual Q-Bio conference started from 2007 is an intimate and dynamic venue that brings together researchers who aim to advance predictive modeling and theoretical understanding of cellular and molecular biological systems in coordination with quantitative experimentation.
On July 21st, the IDF 2023 (International Diabetes Federation Western Pacific Congress 2023)/15th AASD (Asian Association for the Study of Diabetes) scientific meeting took place in Kyoto, Japan. Dr. Huixia Ren was invited to contribute a talk titled “Globally phase-locked alpha and beta cells.” This marked the first in-person meeting since the Covid-19 pandemic.

On June 21th, the 84th American Diabetes Association Annual Meeting was held in San Diego. Dr. Huixia Ren, Dr. Beichen Xie and Yi Yu attended the conference and communicated their research via posters sharing. The ADA’s Scientific Sessions, host to thousands of annual attendees, is the premier diabetes conference providing a platform to delve into the latest advancements in diabetes research, prevention, and care.
2022
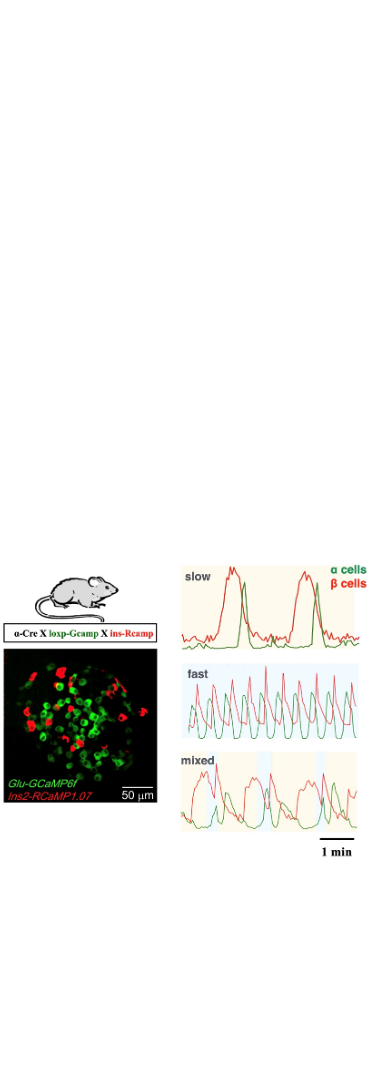
On June 28th, 2022, we released paper « pancreatic α and β cells are globally phase-locked» in Nature Communications(doi:10.1038/s41467-022-31373-6). The Ca2+ modulated pulsatile secretion of glucagon and insulin by pancreatic α and β cells plays a key role in glucose homeostasis. However, how α and β cells coordinate via paracrine interaction to produce various Ca2+ oscillation patterns is still elusive. To address this question, we combined microfluidic device, α and β cells specific labeled transgenic mice, fluorescence imaging and mathematical modelling. We found the oscillations of α and β cells were globally phase-locked. And the activation of α cells displayed a fixed time delay of ~20 s to that of β cells, β cells activated with a tunable delay after the α cells. As a result, the tunable phase shift between α and β cells set the islet oscillation period and pattern in both the model and experiments. Our study highlights the importance of paracrine interaction to generate various islet oscillation patterns. Huixia Ren, Yanjun Li, Chengsheng Han, and Yi Yu mainly contributed to the work. Prof. Chao Tang and Prof. Liangyi Chen supervised the study.
2021
The 57th European Association for the Study of Diabetes Annual Meeting (EASD) of the American Diabetes Association in 2021 was held online from September 27th to October 1st. The conference showcased the research progress in the field of diabetes. Ren Huixia gave an oral presentation “Pancreatic alpha and beta cells are globally phase-locked “in the Keeping the balance in islet secretion session “. During the conference, Professor Anders Tengholm host the discussion session. Presentation: https://www.easd.org/media-centre/home.html#!resources/b-pancreatic-alpha-and-beta-cells-are-globally-phase-locked-b
2018

Prof. Erik Gylfe and Prof. Anders Tengholm from Department of Medical Cell Biology, Uppsala University visited Center for Quantitative Biology (CQB) and Tang Lab from the 9th to the 13th of April, 2018. During their visit, they gave two seminars about pancreatic islet hormone secretion and the mechanisms behind. They also experience Chinese culture and food, and shared with students some “old” stories about the islet study.






